Latest Videos, Blogs, and News
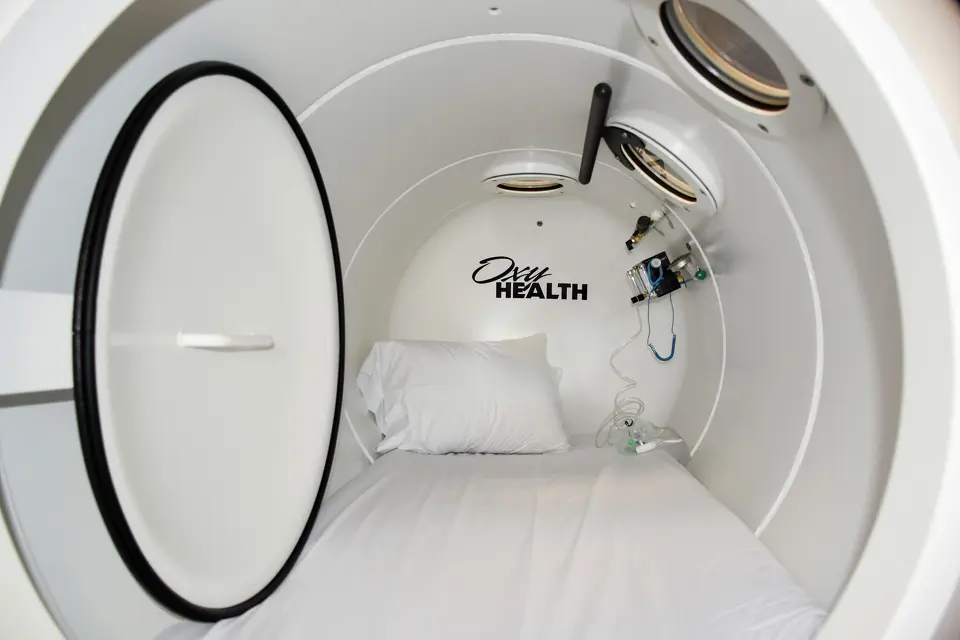
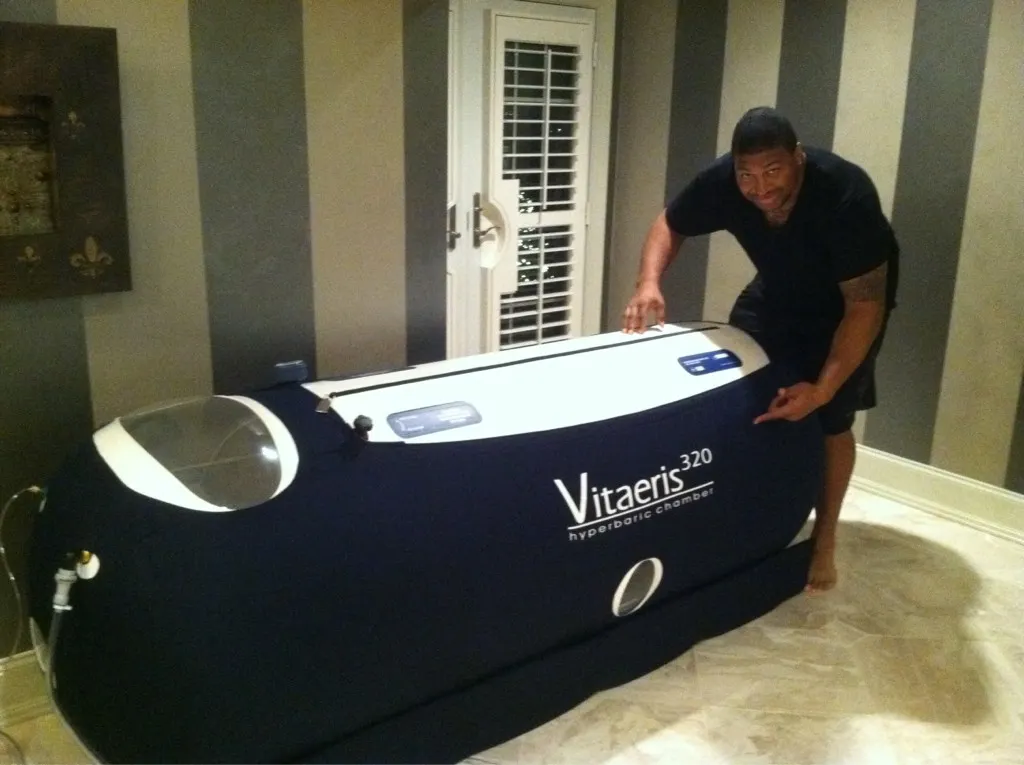
The OxyHealth Resource Center brings together videos, blogs, and news to help you better understand hyperbaric technology. Discover educational content covering the science, benefits, and real-world use of hyperbaric chambers. Whether you’re new to hyperbaric therapy or looking to expand your knowledge, our resources are designed to keep you informed and confident.

1234
123
45
567
Beautiful sunset over the mountains. Nature never fails to amaze us!
Lorem ipsum dolor sit amet consectetur adipisicing elit. Quisquam, quos.

1234
123
45
567
Beautiful sunset over the mountains. Nature never fails to amaze us!

1234
123
45
567
Beautiful sunset over the mountains. Nature never fails to amaze us!

1234
123
45
567
Beautiful sunset over the mountains. Nature never fails to amaze us!

1234
123
45
567
Beautiful sunset over the mountains. Nature never fails to amaze us!

1234
123
45
567
Beautiful sunset over the mountains. Nature never fails to amaze us!

1234
123
45
567
Beautiful sunset over the mountains. Nature never fails to amaze us!
Username...
Loading...
00
Followers
00
Following
Follow